Hepatitis C
Hepatitis C virus is an RNA virus of the flaviviridae family. It develops into a chronic infection in about 70% of patients. The other 30% of patients eliminate the infection naturally.
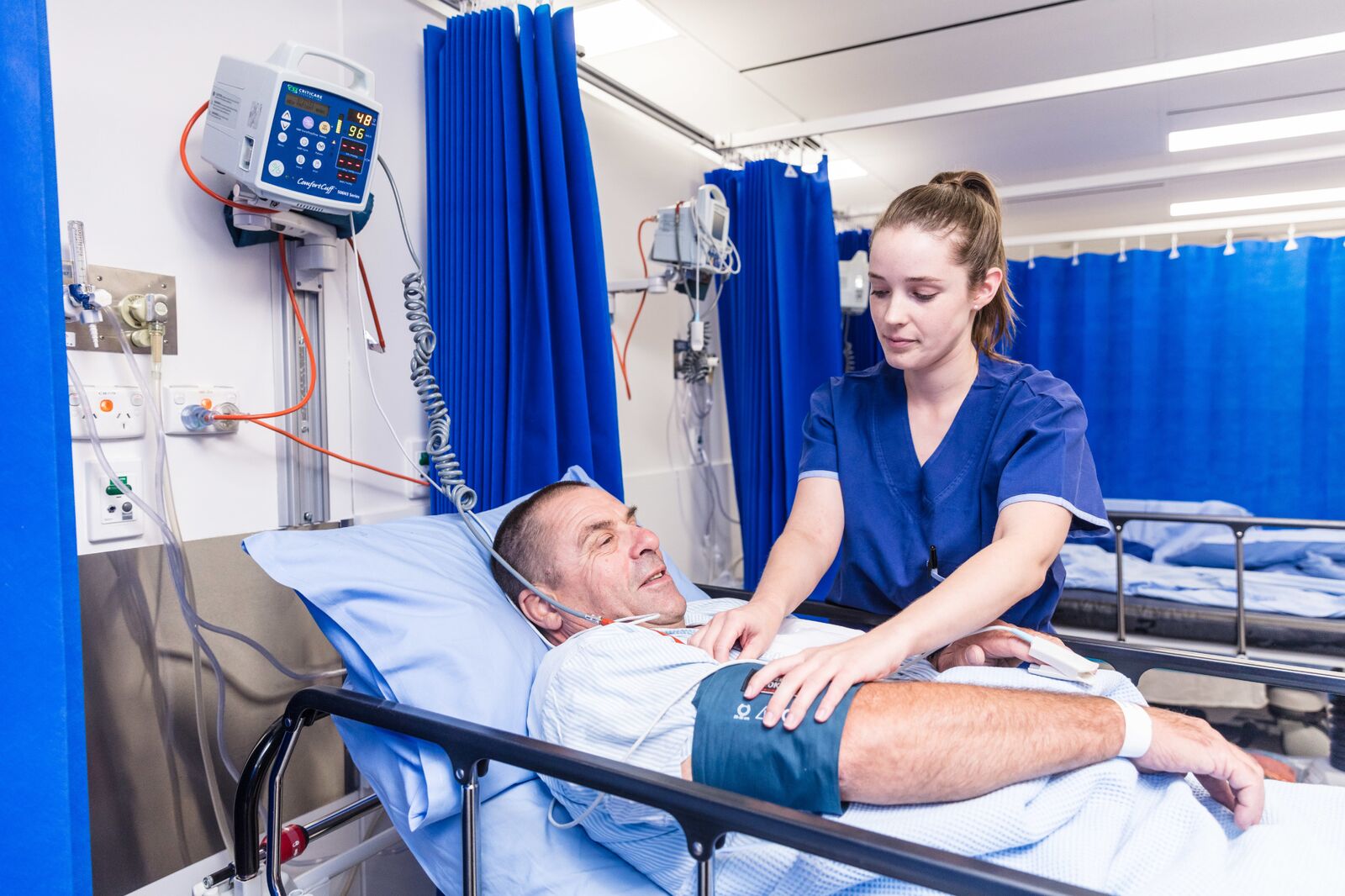
A large pool of undiagnosed hepatitis C patients exists in Australia. Most have acquired HCV through previous intravenous drug use or blood transfusion before 1990. Some cases have no identifiable aetiology. HCV is an RNA virus of the flaviviridae family. It develops into a chronic infection in about 70% of patients. The other 30% of patients eliminate the infection naturally. This is particularly so in children or in adults who have had a mild isolated contact. Transmission risks of HCV are negligible through the birthing process, normal family contacts or sexual activity. Breast feeding is safe. The virus requires blood to blood transmission.
30% of patients are thought to develop chronic liver disease. Cirrhosis supervenes after 20-30 years. Death from hepatoma or liver failure occurs after a further 20 years. Patients may therefore not succumb to HCV until 50 years after infection. Chronic HCV seems to be more debilitating to the host than hepatitis B infection. Tiredness and fatigue are prominent but some patients may feel perfectly well. The ALT is usually elevated in chronic disease but may fluctuate into the normal range. The height of the ALT bears no relationship to the presence or absence of symptoms or to the extent or severity of the liver disease. In other words, a patient can feel perfectly well and still have a HCV cirrhosis with normal liver enzymes. Aggravating factors that shorten the time to cirrhosis are haemochromatosis and alcohol ingestion. Patients who are concurrently infected with hepatitis B generally have a non-replicative hepatitis B state in association with an active hepatitis C. The vice-versa does not usually occur.
Investigations for hepatitis C include LFTs, FBC, genotype strain and quantitative viral load by PCR, and screening for the rare liver diseases of haemochromatosis, Wilson’s disease, alpha-1 antitrypsin deficiency and auto-immune disease. Liver biopsy is essential as the severity of the liver disease bears no relation to the liver enzymes. The biopsy is scored for severity of inflammation and fibrosis. The patient is counselled to minimize alcohol.
Hepatitis C is a curable disease in approximately 50-90% of patients with the latest treatments using pegylated interferon and ribaviron. This requires a once weekly subcutaneous injection and daily oral tablets. The cure rate depends on the genotype strain being carried. In Australia, strain types 1, 2 and 3 are the most common. Genotype 1 strains are the older strains and evolved up to the 1980’s. They are more resistant to therapy and require a 12 month course. Genotypes 2 and 3 have a high cure rate of 80-90% with a 6 month course. They probably arose as mutations during the 1980’s. Patients with a high viral load of greater than 1.2 million viruses per mL are difficult to cure. Other good prognostic points are being female, thin and not cirrhotic.

Polyethylene glycol (PEG) acts a stabilizer to genetically engineered Interferon, one of the body’s natural cytokines and viral killing proteins. Ribavirin is an oral antiviral drug which is active against HCV and inhibits replication. The combination therefore reduces the viral load to allow the Interferon to eliminate the infection. Side-effects are not uncommon, especially fatigue, myalgia, sleeplessness and some emotional irritability and liability. The symptoms are usually tolerable. Some patients may need simple analgesia (Paracetamol), sleeping tablets (Stilnox) or anti-nauseants (Maxalon). The tricyclics (Sinequan) are useful to prevent depression in patients who are low-spirited or who have a history of depression or sleep disorders. Thyroid disease (both hypo and hyperthyroidism) is seen in a small proportion of patients during therapy in whom such problems would have developed in later life. Caution has to be used in patients with psoriasis as this can become worse during therapy. The ALT level is monitored during therapy for consistent normalization. PCR qualitative testing is performed at the half-treatment time mark to assess an adequate viral response with a negative PCR or log 2 reduction in viral load. Long-term virological response is determined by a negative qualitative PCR nine months after cessation of therapy. This is considered to equate to cure of HCV. Patients generally return to a good quality of life in the months after successful cure.